 The checker found no problems in this document. Health care professionals and consumers may report adverse reactions or quality problems they experienced using these devices to MedWatch: The FDA Safety Information and Adverse Event Reporting Program using an online form, regular mail, or FAX.Acrobat Accessibility Report Accessibility Report Filename: RELEASE_75-53153A_Coram_Changing_Injection_Cap_flyer_WCAG.pdf Report created by: Lisa Comfort, Graphic designer (Contractor), Organization: CVS Health® (Arrow International’s parent company) for a refund.Ĭustomers with questions should call Teleflex customer service at 86 or email Additional Resources: 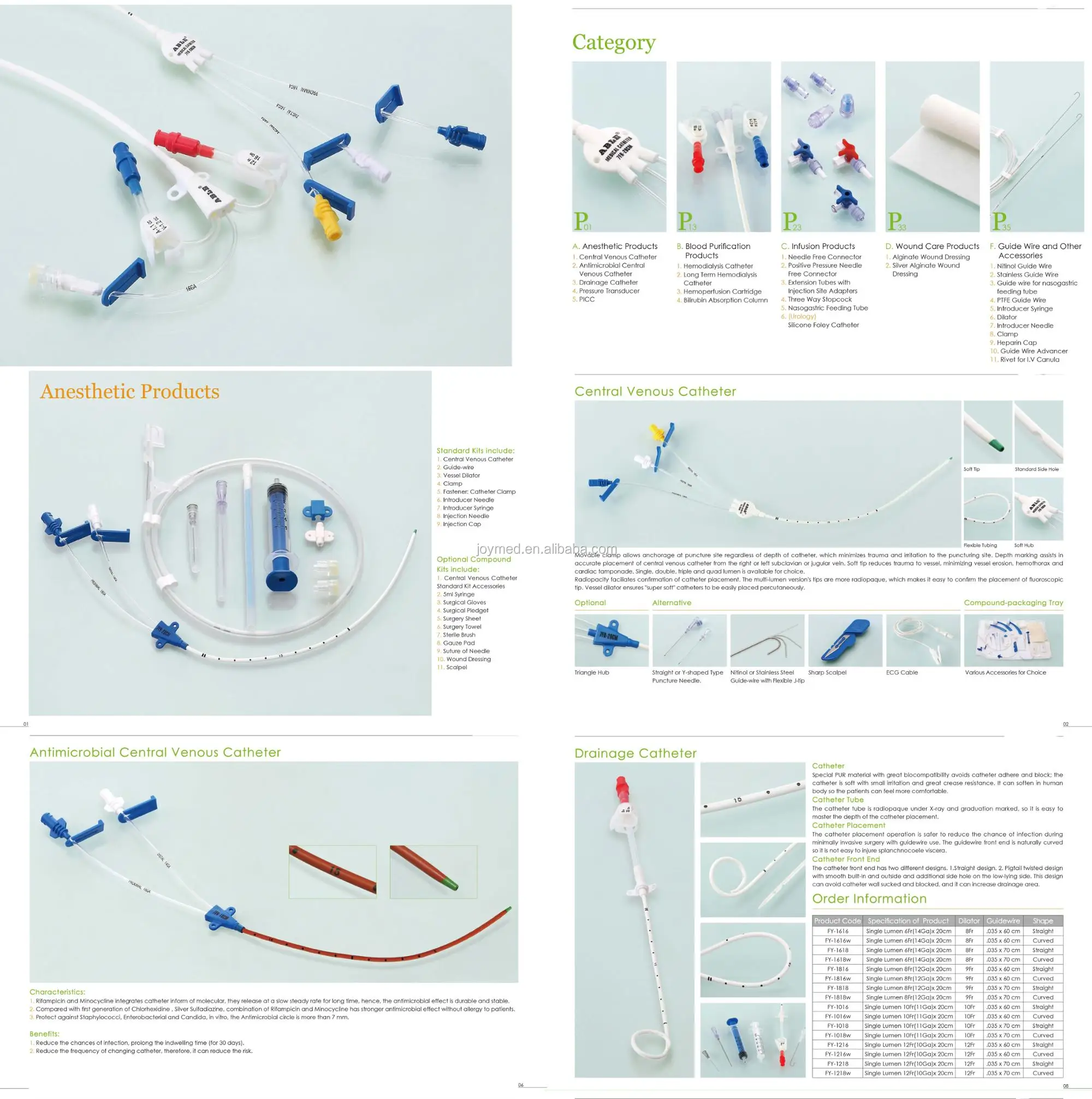
Return affected products to Teleflex Inc.Stop using and distributing affected products.The letter recommended the following actions: On October 27, 2022, Teleflex and Arrow International, LLC sent customers an Urgent Medical Device Recall letter. Distributors of the Arrow MAC Two-Lumen Central Venous Access Kits Arrow Pressure Injectable Arrowg+ard Blue Plus Three-Lumen Central Venous Catheter (CVC) Kits.Health care personnel providing care using the Arrow MAC Two-Lumen Central Venous Access Kits Arrow Pressure Injectable Arrowg+ard Blue Plus Three-Lumen Central Venous Catheter (CVC) Kits.People who receive care using the Arrow MAC Two-Lumen Central Venous Access Kits Arrow Pressure Injectable Arrowg+ard Blue Plus Three-Lumen Central Venous Catheter (CVC) Kits.Teleflex/Arrow International LLC has reported no injuries or deaths related to this issue. 5,6,10 Knowledge of strategies to prevent catheter-related infec-tions is essential. Use of affected devices may cause bleeding, fluid leakage, delayed treatment, infection, air in the blood vessels (air embolism), other serious injuries or death. Central lineassociated bloodstream infection (CLABSI) is linked to increased mortality of up to 25 and greater than 1 billion in associated costs. Teleflex and their subsidiary Arrow International, LLC are recalling the Arrow MAC Two-Lumen Central Venous Access Kits Arrow Pressure Injectable Arrowg+ard Blue Plus Three-Lumen Central Venous Catheter (CVC) Kits for the risk of a cross-lumen leak caused by inadequate connections between the top and bottom housings of the Micro Clave Clear Connectors included in the kits. 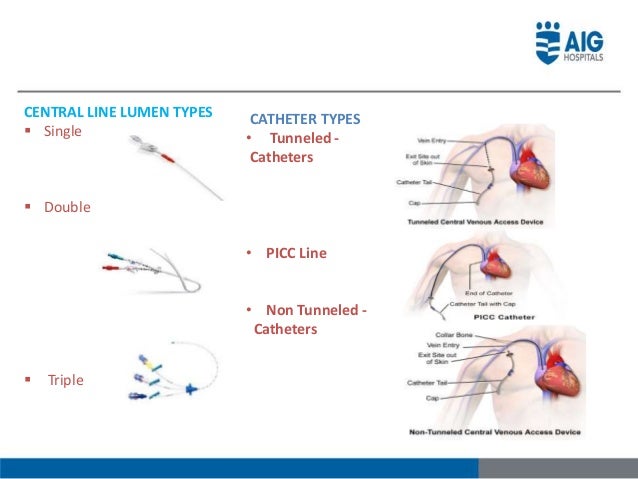
It is not intended to be used as a treatment for existing infections. The Arrow Pressure Injectable Arrowg+ard Blue Plus Three-Lumen CVC Kit is intended to provide short-term (less than 30 days) protection against catheter-related bloodstream infections. CVCs range widely in diameter, from 3-Fr single lumen PICCs to 7-Fr 5-lumen CVCs to 15-Fr double-lumen dialysis catheters. This may vary by manufacturer, but typically each lumen of the catheter is a different color, its own separate line, and used for designated purposes. The Arrow MAC Two-Lumen Central Venous Access Kit is intended to permit short term (less than 30 days) venous access and catheter introduction to the central circulation. When I worked in the ICU, patients were often receiving multiple medications and fluids at once, so a triple lumen central catheter, and knowing the function of each port, was essential. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |


 RSS Feed
RSS Feed
